Organisation name: Clinical Research Excellence Foundation (formerly known as ClinverseEdge)

28 January, 2026
Author : Dr Vijaykumar Gawali
Topic : ROLE OF A CRC IN PATIENT RECRUITMENT
Definition & Importance
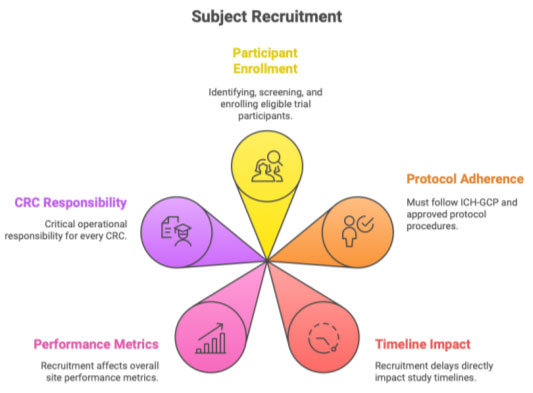
Patient recruitment is the process of identifying, screening, and enrolling eligible participants into a clinical trial following ICH-GCP, protocol requirements, and EC-approved procedures.
For a CRC, recruitment is one of the most critical responsibilities because delays in recruitment directly impact study timelines and site performance.
Responsibilities of the CRC in Recruitment
A CRC plays a central, operational, and compliance-focused role in recruitment:
A. Pre-Recruitment Activities (Before EC Approval)
These activities are allowed only if no direct patient contact occurs:
Feasibility Assessment
- Review hospital patient load for the study indication.
- Extract data from:
- HIS (Hospital Information System)
- OPD/IPD registers
- Specialty departmental registries
- Diagnostic center data
- Prepare a potential patient pool report.
Drafting Recruitment Strategy for EC Submission
CRC assists the PI in drafting:
- In-house recruitment methods (posters, standees, brochures)
- Multimedia recruitment (hospital website, sms system, whatsapp broadcast per policy)
- Newspaper and print media ads
- Referral pathway from internal departments
➤ All recruitment materials must be EC-approved before use.
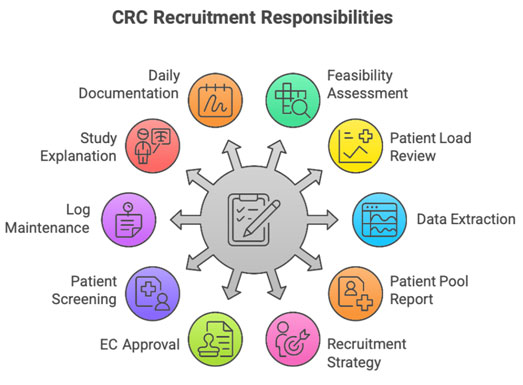
B. Recruitment Activities After EC Approval
OPD/IPD Screening with the PI
CRC sits in OPD with the investigator to:
- identify potentially eligible patients
- verify inclusion/exclusion criteria
- maintain pre-screening log
- prepare patients for detailed screening
Explaining the Study
CRC supports the PI by:
- arranging a private room for discussion
- providing EC-approved ICF
- answering procedural questions
- scheduling screening visits
Documenting All Recruitment Activity
- Pre-screening log
- Screening log
- Referral log
- Advertising/IEC-approved materials
- Summary of daily OPD screening activity
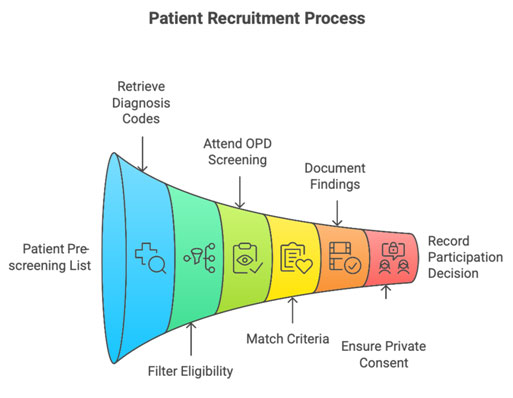
Step-by-Step Recruitment Workflow
Step 1: Create Patient Pre-Screening List
From HIS/departmental registries:
- Retrieve diagnosis codes
- Filter appropriate age groups
- Prepare list for PI review
Step 2: OPD/Clinic Screening
- Attend OPD with PI
- Match patients to eligibility criteria
- Document in pre-screening log
Step 3: Explain Study (after PI introduces the study)
- Ensure privacy
- Provide ICF
- Use non-coercive language
- Answer questions
Step 4: Document Decision
- Consented? → Proceed to screening
- Not interested? → Record reason in pre-screen log
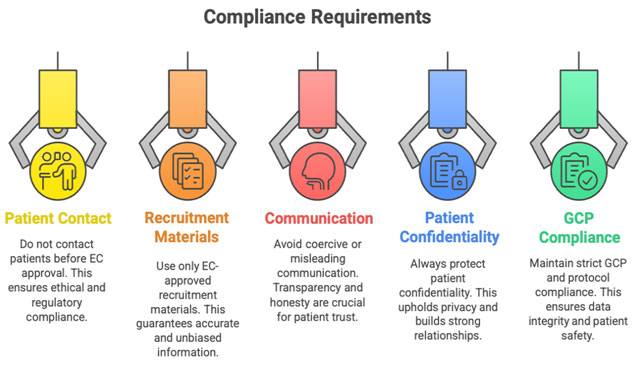
Compliance Requirements
- No patient may be contacted before EC approval.
- No unapproved recruitment materials may be used.
- No coercion, pressure, or misleading information.
- Protect patient confidentiality at all times.